About Avid Bioservices
Avid Bioservices, Inc. operates as a contract development and manufacturing organization (‘CDMO’) that provides a comprehensive range of services from process development to Current Good Manufacturing Practices (‘CGMP’) clinical and commercial manufacturing of biologics for the biotechnology and biopharmaceutical industries.
The company’s services include clinical and commercial drug substance manufacturing, bulk packaging, release and stability testing and regulatory submissions support. The company also provides a variety of process development services, including upstream and downstream development and optimization, analytical method development, cell line development, testing and characterization.
The company manufactures CGMP pharmaceutical-grade products for its customers.
Business Strategy
The company continues to execute on a growth strategy that seeks to align with the growth of the biopharmaceutical drug substance contract services market.
Growth Strategy

The company has a significant opportunity to continue to drive organic growth by leveraging the company’s strengths, broadening the company’s capabilities, increasing the company’s capacity and improving the company’s market visibility through the following strategies: diversifying customer base; expanding service offerings; expanding process development capabilities; expanding manufacturing footprint and enhancing efficiencies; reinvesting in equipment and facilities; and exploring and investing in strategic opportunities.
Regulatory Matters
The company has been audited by several regulatory agencies, including the FDA, the European Medicines Agency (‘EMA’), the Brazilian Health Surveillance Agency (‘ANVISA’), the Canadian Health Authority (‘Health Canada’), the California Department of Health, and the Australian Department of Health.
The company is required to comply with the regulatory requirements of various local, state, national and international regulatory bodies having jurisdiction in the countries or localities where the company manufactures products or where its customers’ products are distributed. In particular, the company is subject to laws and regulations concerning research and development, testing, manufacturing processes, equipment and facilities, including compliance with CGMPs, labeling and distribution, import and export, and product registration and listing. As a result, the company’s facilities are subject to regulation by the FDA, as well as regulatory bodies of other jurisdictions where the company’s customers have marketing approval for their products, including but not limited to, the EMA, ANVISA, Health Canada, and the Australian Department of Health.
Intellectual Property
Avid Bioservices is a registered trademark of the company.
History
The company was founded in 1981. It was incorporated in California in 1981 and reincorporated in the state of Delaware in 1996. The company was formerly known as Peregrine Pharmaceuticals, Inc. and changed its name to Avid Bioservices, Inc. in 2018.

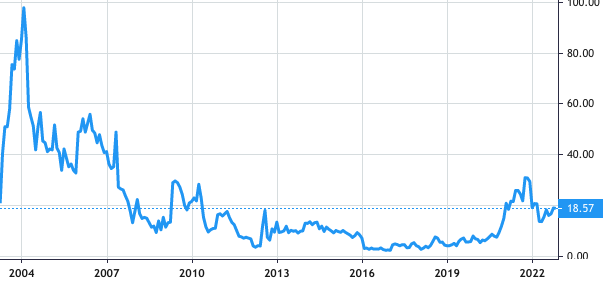
 Stock Value
Stock Value