About Daxor
Daxor Corporation operates as a medical device manufacturing company. The company focuses on the creation and development of the BVA-100 Blood Volume Analyzer, an instrument that measures human blood volume. This instrument is used in conjunction with Volumex, a single-use radiopharmaceutical diagnostic injection and collection kit.

The company also offers cryobanking services for blood storage through a wholly owned subsidiary, Scientific Medical Systems. It also owns the Daxor Oak Ridge Operations (DORO) facility in Oak Ridge, Tennessee, which manufactures, tests, and develops next-generation models of the BVA-100.
BVA-100 BLOOD VOLUME ANALYZER
The BVA-100 is a Clinical Laboratory Improvement Act (CLIA)-rated medium complexity instrument that can measure blood volume with 98% accuracy within a 60 to 90 minute time frame. The BVA-100 is used to diagnose and treat patients with heart failure, kidney failure, hypertension and syncope, and to aid in fluid and blood transfusion management in the critical care unit. The BVA-100 also has been used to aid in the diagnosis and treatment of disorders of red blood cell volume, including polycythemia and anemia, and to aid in pre-surgical evaluation of red blood cell volume. It may also be possible to use the BVA-100 to manage kidney dialysis, ultrafiltration, and blood optimization for elective surgery.
The company’s patented injection and collection kit (Volumex) utilizes Albumin I-131, a classic tracer used in blood volume measurement. The BVA-100 software automatically calculates the blood volume, evaluates the statistical reliability of the measurement, and compares the results to the known predicted norm, which is a function of the patient’s height, weight and gender.
Scientific Medical Systems

Scientific Medical Systems engages in cryobanking of human blood. Idant Laboratories (Idant), a division of Scientific Medical Systems, provides semen banking services. The company has received a trademark for a proposed program of Quality Assured Blood (QAB).
The company also has trademarked its Blood Optimization Program (BOP) for maximizing blood safety during surgery. The BOP uses a combination of blood volume measurement and pre-surgical treatment of blood volume deficits to maximize patient outcomes following surgery. The company has applied for and received trademark protection for the BOP name.
Idant Semen (Sperm) Banking
Idant provides frozen semen services to physicians worldwide. It holds approximately 50,000 human semen units in long-term storage at its central New York City facility. The company stores semen from a large cross-section of anonymous donors and is able to offer semen from donors with varying physical characteristics that meet its clients’ needs. The company maintains a complete physical description of each donor on file and, when needed, can match multiple physical characteristics and other desired special characteristics to those of the sterile father. The company also provides cryostorage of semen for later personal use.
Research and Development
As of December 31, 2011, the company has spent $2,705,952 on research and development.
Patents
The company owns a separate patent on its Volumex injection kit which expires in 2016. It also received a European patent covering 12 countries. The blood bank has received two trademarks: one is for Quality Assured Blood and the other is for the BOP. The company has applied for and received trademark protection for the BOP name.
Regulation
As a registered medical device manufacturer, the company is subject to periodic inspection by the Food and Drug Administration (FDA) of manufacturing facilities, production records, product design records and complaint files. The company conducts regular internal audits to ensure compliance with all good manufacturing (GMP) regulations. The New York State Department of Health regulates the company’s Idant Semen and Blood Banks within New York State. These facilities are licensed and annually inspected by the New York State Department of Health.
History
Daxor Corporation was founded in 1970. The company was incorporated in New York state as Iatric Corporation in May 1971 and changed its name to Idant Corporation in October 1971. In 1973, the company’s name was changed to Daxor Corporation.

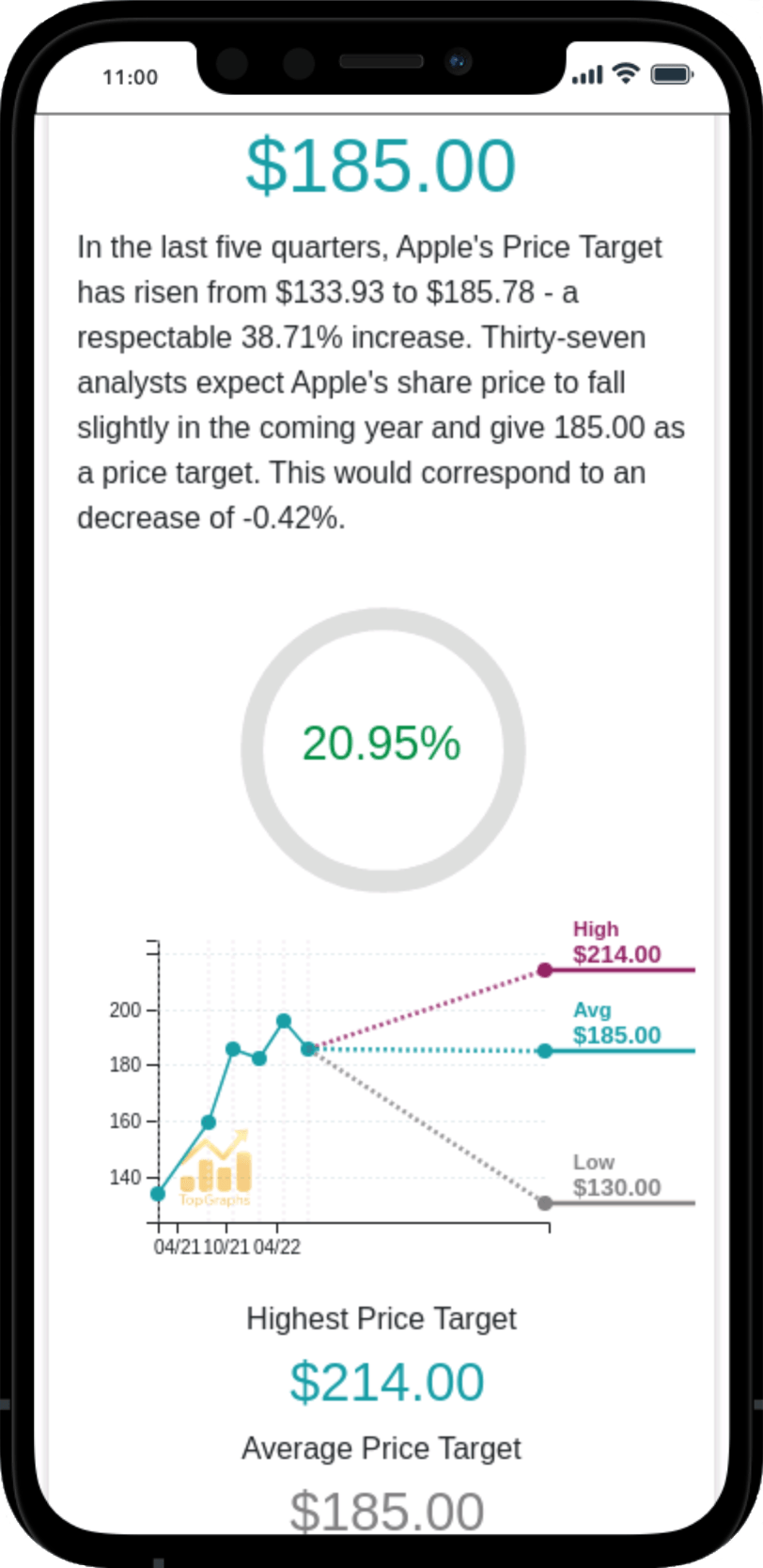
 Stock Value
Stock Value