About Enovis
Enovis Corporation (Enovis) operates as a medical technology company.

The company is focused on developing clinically differentiated solutions that generate measurably better patient outcomes and transform workflows by manufacturing, and distributing high-quality medical devices with a broad range of products used for reconstructive surgery, rehabilitation, pain management and physical therapy. The company’s products address the continuum of patient care from injury prevention to rehabilitation after surgery or injury or from degenerative disease, enabling people to regain or maintain their natural motion. The company seeks to leverage its Enovis Growth eXcellence business system (‘EGX’), a set of tools, processes, and culture, to continuously improve the company’s ability to enable great patient outcomes and to drive and fuel growth.
Segments
The company operates through the Prevention & Recovery and Reconstructive segments. The company develops, manufactures and distributes high-quality medical devices and services across the continuum of patient care from injury prevention to joint replacement to rehabilitation after surgery, injury or from degenerative disease, enabling people to regain or maintain their natural motion. The company reaches a diverse customer base through multiple distribution channels, that include both independent distributors and direct salespeople, and provide a wide range of medical devices and related products to orthopedic specialists and other healthcare professionals operating in a variety of patient treatment settings and to retail consumers.
Prevention & Recovery
The company’s Prevention & Recovery segment includes products that are used by orthopedic specialists, surgeons, primary care physicians, pain management specialists, physical therapists, podiatrists, chiropractors, athletic trainers and other healthcare professionals to treat patients with musculoskeletal conditions resulting from degenerative diseases, deformities, traumatic events and sports-related injuries. In addition, many of the company’s non-surgical medical devices and related accessories are used by athletes and patients for injury prevention and at-home physical therapy treatment. The company’s Prevention & Recovery product lines include rigid and soft orthopedic bracing, hot and cold therapy, bone growth stimulators, vascular therapy systems and compression garments, therapeutic shoes and inserts, electrical stimulators used for pain management and physical therapy products.

Reconstructive
The company’s Reconstructive segment is an innovation-driven leader offering a comprehensive suite of reconstructive joint products for the hip, knee, shoulder, elbow, foot, ankle, and finger and surgical productivity tools.
Competition
Key competitors for the company’s Prevention & Recovery segment include Össur and Breg, Inc.
Key competitors for the company’s Reconstructive segment include Stryker, Zimmer Biomet, and DePuy Synthes, the medical device business within Johnson & Johnson.
International Operations
The company’s principal market for the company’s Prevention & Recovery and Reconstructive segments outside the U.S. is Europe. For the year ended December 31, 2023, approximately 32% of the company’s Net sales were derived from operations outside the U.S., the majority of which is in Europe with the remaining portion mostly in the Asia-Pacific region.
Seasonality
The company’s sales typically peak in the fourth quarter (year ended December 2023), however, the business impact caused by the COVID-19 pandemic has distorted the effects of historical seasonality patterns.
Regulatory Environment
In the United States, the company’s products generally are subject to regulation by the Food and Drug Administration (the ‘FDA’) as medical devices pursuant to the Federal Food Drug and Cosmetic Act (the ‘FDCA’).
To obtain 510(k) clearance, the company must submit to the FDA a premarket notification submission demonstrating that the proposed device is ‘substantially equivalent’ to a predicate marketed device.
The company is actively working towards obtaining MDR-certification with the company’s notified body.
Healthcare providers that prescribe the company’s products and from which the company obtains patient health information are subject to privacy and security requirements under HIPAA, as are the company in certain circumstances.
History
Enovis Corporation was founded in 1995.
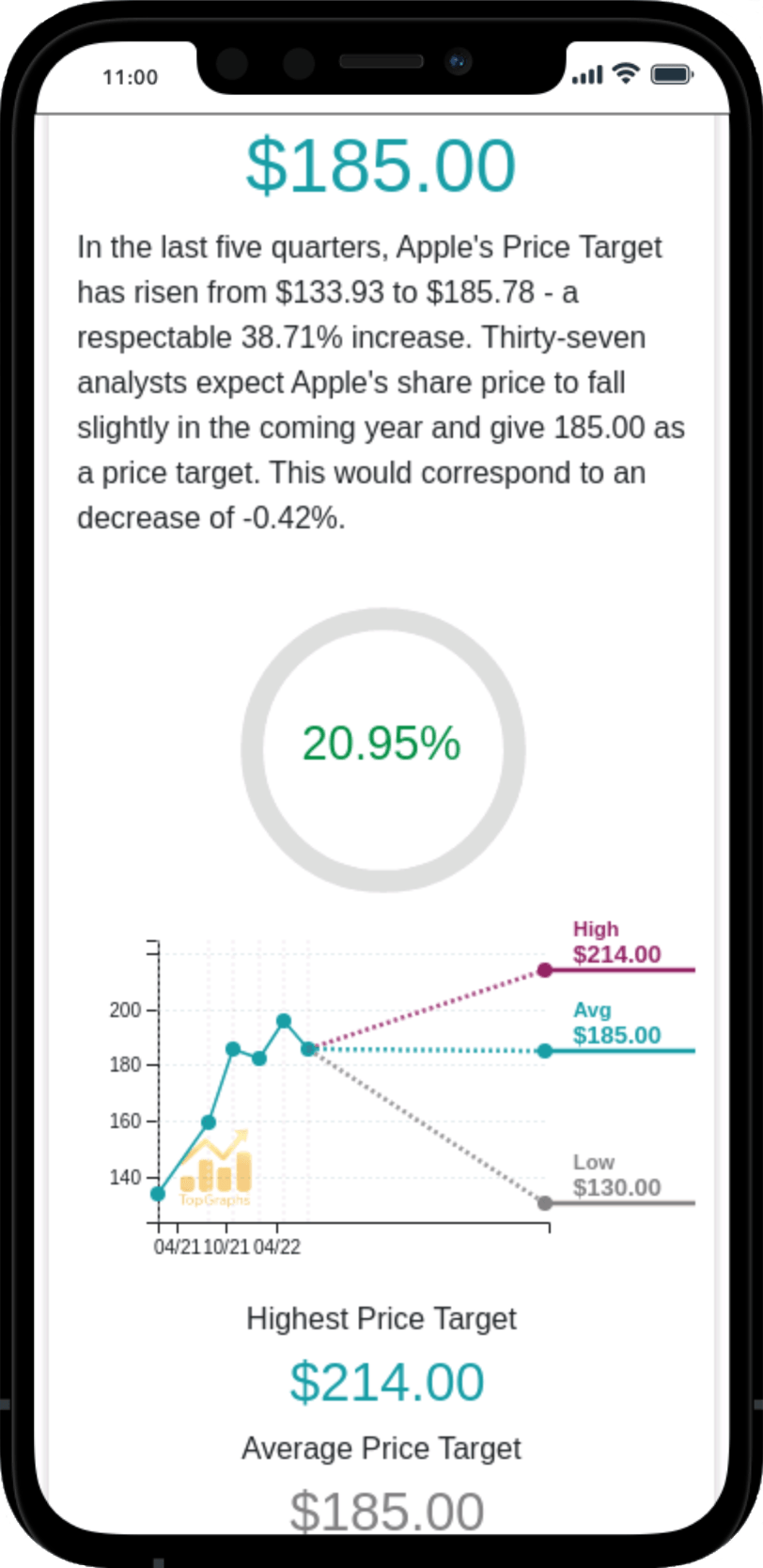
 Stock Value
Stock Value